Researchers at the U.S. Department of Energy’s Oak Ridge National Laboratory (ORNL) have discovered a new way to design superionic polymer electrolytes that could transform solid-state batteries and other energy storage technologies. Their work offers a promising route toward safer, faster-charging, and more reliable energy systems for the future.
The team showed that by precisely controlling the chemical composition of a lithium salt–based polymer, they could create a material that allows ions to move at extremely high speeds. This breakthrough could help overcome one of the biggest challenges in battery science—slow ion transport in solid electrolytes.
“Researchers around the world are focusing on unlocking the potential of polymer electrolytes because they have a lot of advantages over conventional liquid electrolytes,” said Catalin Gainaru of ORNL’s Chemical Sciences Division. “Achieving fast ion transport has always been a major challenge, but our recent research shows this may no longer be the case.”
Batteries work through the movement of ions between two electrodes, the cathode and anode, through an electrolyte. For efficient charging and discharging, these ions must move quickly and smoothly. While liquid electrolytes are commonly used today, they come with safety risks and limitations. Solid-state batteries offer a safer and more compact alternative, but improving ion movement inside solid materials has been difficult.
READ ALSO: https://modernmechanics24.com/post/nrd-nuclear-battery-100-year-power/
Traditionally, ceramic electrolytes have shown excellent ion transport and are called superionic materials. However, they are brittle, difficult to process, and do not integrate well into flexible battery designs. ORNL’s new polymer-based approach overcomes these limitations by combining high ion mobility with mechanical flexibility.
The researchers demonstrated that ions in their polymer system can move up to 10 billion times faster than the surrounding material. This creates a “superionic” state within a flexible polymer matrix, offering the advantages of both ceramics and liquids without their drawbacks.
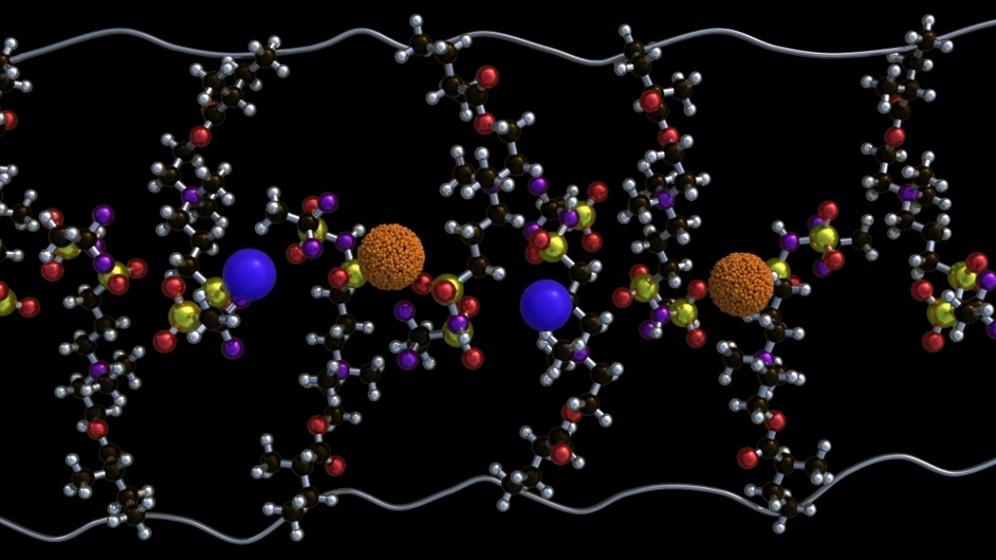
Polymers are long-chain molecules made of repeating units, commonly found in plastics. In this study, the ORNL team engineered a special polymer containing polar segments that attract lithium salts and enhance ion mobility. This design allows ions to move more freely and efficiently across the material.
The research, published in Materials Today, was conducted under the DOE Energy Frontier Research Center known as FaCT (Fast and Cooperative Ion Transport in Polymer-Based Materials). The goal of the center is to fundamentally redesign how ion transport is achieved in polymers.
WATCH ALSO: https://modernmechanics24.com/post/buildroid-ai-expands-robotics-platform/
“We developed a very special polymer in which the segments self-organize to provide a high mobility path for the ions to move through,” said Tomonori Saito, an ORNL distinguished researcher.
A key innovation in the study was the use of zwitterions—molecular groups that carry both positive and negative charges. By carefully adjusting the amount of these groups, researchers tuned how ions organize within the polymer. At an optimal level of around 80% functionalization, the ions form connected pathways that enable smooth and efficient transport.
The behavior of ions in the material is compared to conversations at a gathering. Small isolated “clusters” form at first, then merge into a smooth flow of interaction. However, if too many zwitterions are added, the structure becomes disordered, slowing ion movement again. Finding the right balance was crucial to achieving high performance.
READ ALSO: https://modernmechanics24.com/post/us-200m-mq-4c-triton-vanishes-hormuz/
Looking ahead, the research team plans to further study the molecular mechanisms behind this behavior using advanced modeling, AI-driven chemistry, and supercomputing resources. Experiments at ORNL’s Spallation Neutron Source will also help observe ion interactions at the atomic level.
While solid-state batteries are a major application, the potential uses extend far beyond. Flow batteries, fuel cells, grid storage systems, and other energy technologies could all benefit from this discovery.
As Saito noted, “Anything that needs an impermeable barrier layer but allows ions to move freely is a potential application.”