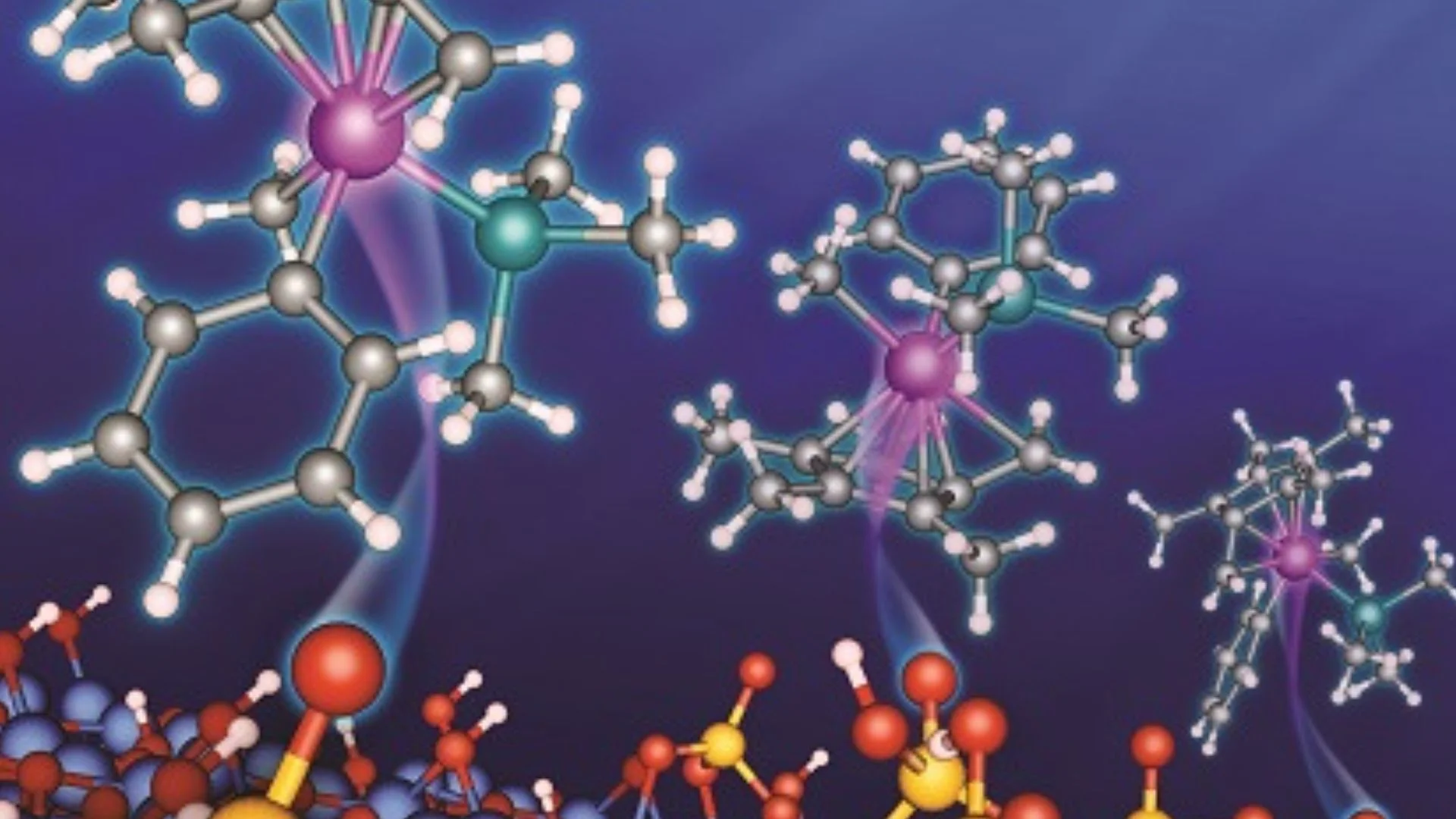
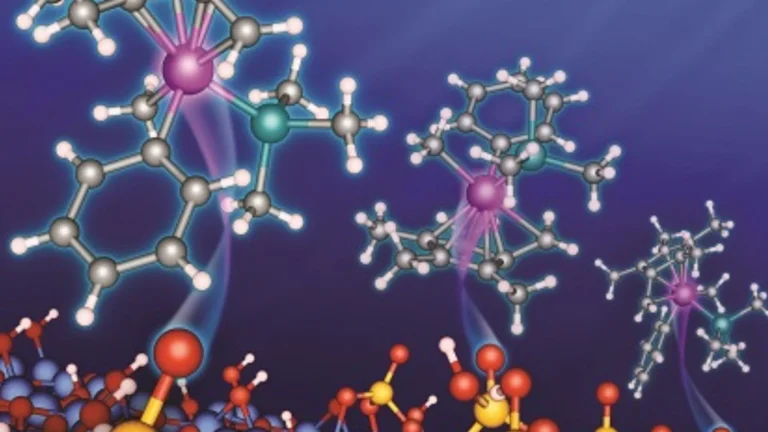
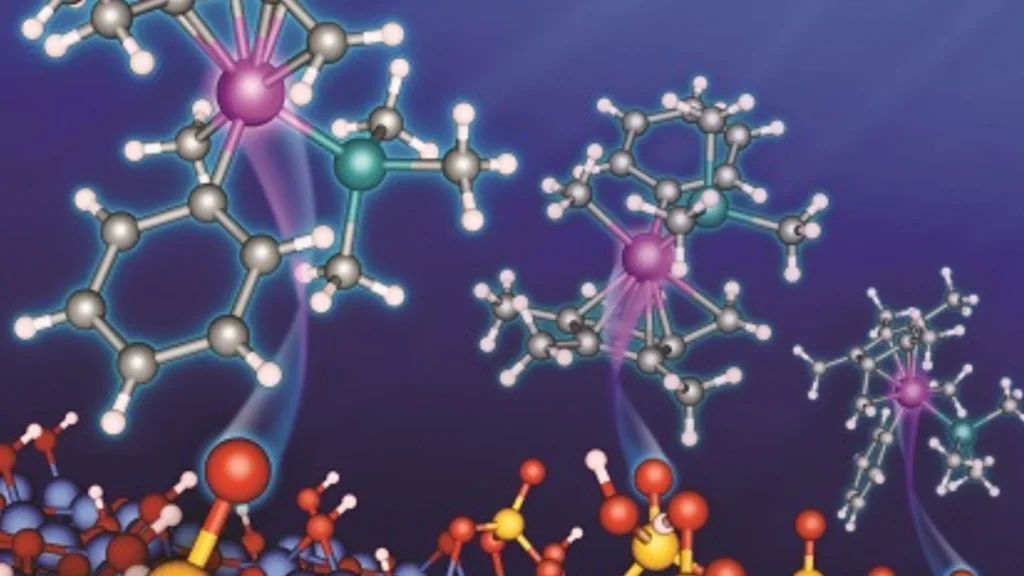
Researchers from the University of Jyväskylä have introduced a new catalyst modeling approach that connects atomic-level chemistry with full industrial reactor performance.
The study shows how advanced computational tools could improve the design of catalysts used in clean hydrogen production, carbon dioxide conversion, and other important chemical processes.
The research, published in ACS Catalysis, focuses on multiscale modeling, a method that combines simulations across different scientific scales.
Scientists say the approach can help industries better predict how catalysts behave under real operating conditions, improving both efficiency and reliability.
Catalysts are materials that speed up chemical reactions without being consumed in the process. They are essential in industries such as fuel production, energy generation, chemical manufacturing, and environmental technology. Modern clean-energy systems, including hydrogen and carbon capture technologies, also rely heavily on efficient catalysts.
READ ALSO: https://modernmechanics24.com/post/wireless-brain-implant-restores-vision/
However, designing better catalysts has remained a major challenge because chemical reactions occur across multiple scales simultaneously. While atomic interactions determine the basic behavior of a catalyst, larger factors such as temperature, pressure, solvents, and reactor conditions also influence overall performance. Researchers say traditional modeling methods often struggle to capture this full complexity.
The new study explains how different computational techniques can work together to solve this problem. The researchers reviewed methods ranging from atomic-level electronic structure calculations to large-scale industrial reaction simulations.
One of the key techniques discussed in the study is Density Functional Theory(DFT). This method allows scientists to examine how atoms and electrons interact inside materials. Researchers use DFT to predict how catalytic surfaces behave during chemical reactions and to identify promising materials for industrial use.
WATCH ALSO: https://modernmechanics24.com/post/american-nextgen-ejection-seat-92-survivability/
According to the research team, atomic-level calculations alone are not enough to explain how catalysts perform in real industrial systems. Factors such as solvents, electrical potentials, and changing operating conditions can strongly influence reactions. By integrating these conditions into simulations, scientists can produce more accurate and realistic predictions.
The researchers say this is especially important for clean-energy technologies. Catalysts play a major role in hydrogen production, carbon dioxide conversion, biomass processing, fuel refining, and emission reduction systems. Better catalyst designs could reduce industrial energy consumption and support more sustainable manufacturing processes.
The study also highlights the growing importance of electrocatalysis, in which electricity drives chemical reactions. Electrocatalytic systems are important for batteries, hydrogen production, and carbon capture technologies. Scientists believe improved catalyst models could help increase the efficiency of these systems.
Researchers noted that advances in computing power and software development are making it easier to simulate highly complex reactions. Earlier computational systems often struggled to connect microscopic chemical behavior with large-scale industrial performance. Improved algorithms and modern computing tools are now helping bridge that gap.
READ ALSO: https://modernmechanics24.com/post/floating-ai-data-centers-silicon-valley/
Academy Research Fellow Minttu Smith said realistic reaction conditions must be included at every stage of modeling. She explained that simulations may fail to match laboratory results if important operating conditions are ignored.
Professor Karoliina Honkala said multiscale modeling can describe the full complexity of catalytic reactions when different methods are carefully combined. She added that understanding both the theoretical calculations and the chemistry behind the reactions is essential for producing reliable predictions.
Researchers believe continued progress in computational chemistry and digital research tools could help industries design cleaner and more efficient chemical technologies in the future.