Researchers at ETH Zurich believe the future of bone repair could lie in something surprisingly soft,a hydrogel.
The research team is led by Professor Xiao-Hua Qin, a biomaterials expert at ETH Zurich, along with Professor Ralph Müller.
A broken bone from a skiing accident usually heals on its own. But when the fracture is severe, or a bone tumor must be removed, doctors often need to insert an implant. These implants help the bone heal.
Surgeons commonly use autografts or metal and ceramic implants. Autografts are bone grafts taken from the patient’s own body. While effective, they require a second surgery.
Metal implants, on the other hand, are very strong but often too rigid. Over time, they may loosen and cause problems.
But the research team now wants to create a bone implant that works with the body’s natural healing process rather than against it.
Professor Qin says, “For proper healing, it is vital that biology is incorporated into the repair process.” She explains that bone is not a solid block. It is a complex organ filled with tiny tunnels and cavities. Different types of cells must enter the implant before new bone can form.
READ ALSO: https://modernmechanics24.com/post/bio-inspired-robotic-wing-underwate/
This insight led the team to develop a novel hydrogel. The study was recently published in the journal Advanced Materials.
When a bone breaks, the body does not immediately create a hard bone. In the first few days, a soft structure forms. A bruise, known as a haematoma, develops at the fracture site. This soft, permeable material allows immune and repair cells to enter the area. Nutrients also flow through easily.
A network of fibrin binds the cells together. With time, this soft structure slowly transforms into hard bone. The new hydrogel is designed to copy this natural process.
The material is made of 97 percent water and 3 percent biocompatible polymer. It feels like jelly. But scientists found a way to control and shape it with extreme precision.
To make the hydrogel solid, researchers added two special molecules. One molecule links the polymer chains together. The other reacts when exposed to light.
When a laser beam hits the hydrogel, the molecules instantly connect the polymer threads. The exposed areas become solid immediately. The parts that are not exposed remain soft and can later be washed away.
Wanwan Qiu, a former doctoral student in the team, developed the connecting molecule. She says, “It enables rapid structuring of hydrogels in the sub-micrometre range.”
READ ALSO: https://modernmechanics24.com/post/uk-awards-leonardo-1-3b-defense-deal/
Using this technology, scientists can print tiny structures into the hydrogel. The details can be as small as 500 nanometres. That is far thinner than a human hair.
Professor Qin says, “With our newly developed connecting molecule, we can structure the hydrogel in a stable and extremely fine manner.” She adds, “We can also produce it at high writing speeds of up to 400 millimetres per second. That’s a new world record.”
In their study, the team created complex hydrogel structures that closely resemble real bone. They used medical imaging data as a template.
Natural bone contains an intricate network of channels filled with fluid. These channels are extremely small, often only nanometres thick. Professor Qin explains, “A piece of bone the size of a dice contains 74 kilometres of tunnels.”
For comparison, the famous Gotthard Base Tunnel is 54 kilometres long. That shows how complex bone truly is.
WATCH: https://modernmechanics24.com/post/ai-platform-powers-research-robot/
By recreating this fine network within the hydrogel, researchers hope to provide bone-forming cells with the ideal environment to grow.
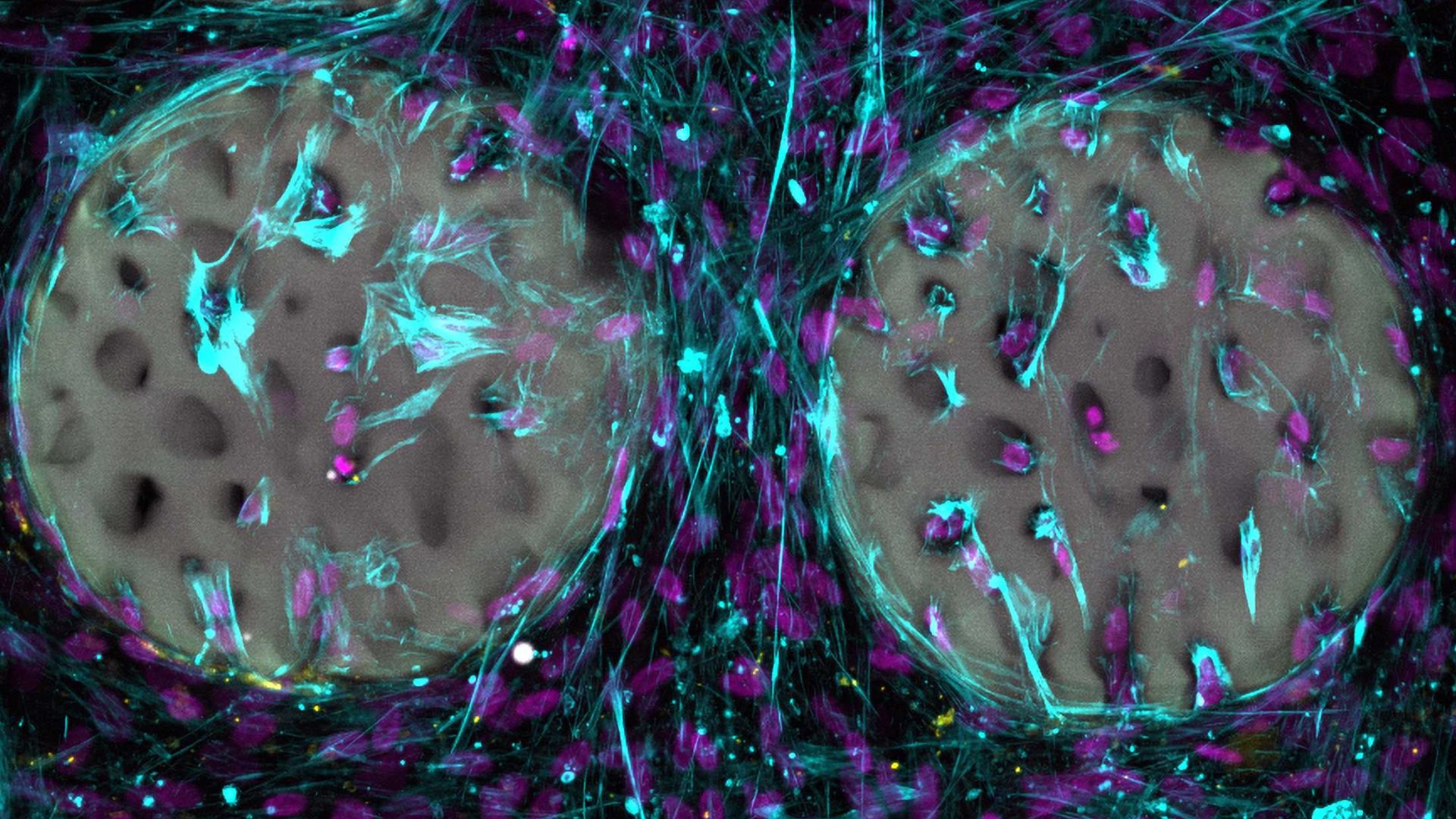
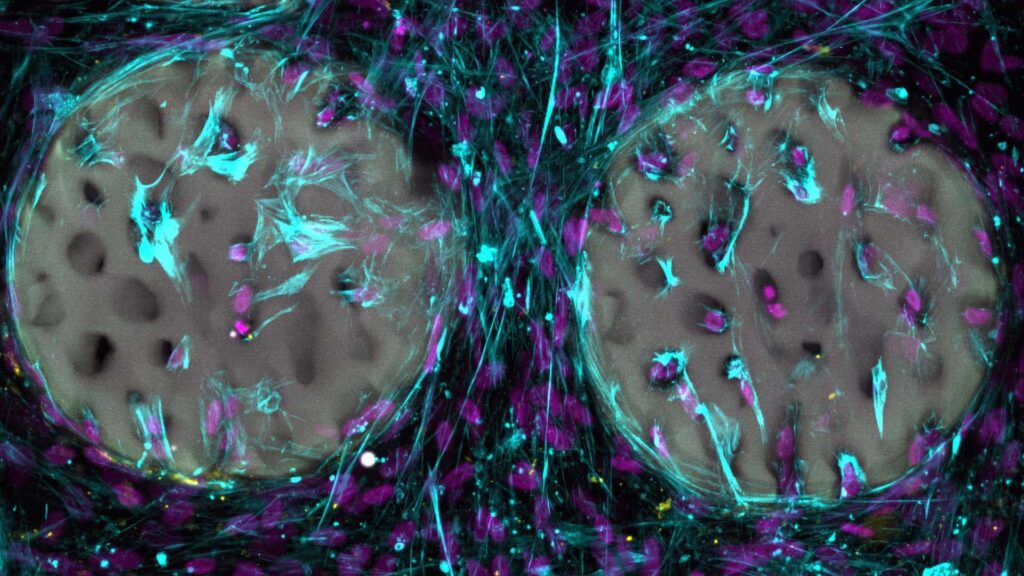
So far, the hydrogel has only been tested in laboratory conditions. The results are encouraging.
Bone-forming cells quickly colonised the structured hydrogel. They began producing collagen, a key building block of bone. The material also proved to be biocompatible. It did not harm the cells.
The researchers have already patented the base material. They now plan to work with the medical industry to further develop it.
READ ALSO: https://modernmechanics24.com/post/building-resilient-plant-cell-by-cell/
The next step involves animal testing. The team will collaborate with the AO Research Institute Davos to study how the hydrogel behaves in living organisms. They want to see if it supports cell migration and restores bone strength over time.
Hydrogel-based implants could one day replace traditional metal and bone graft implants. Because the material dissolves gradually in the body, it may eliminate the need for additional surgery. It could also allow for personalised implants designed from a patient’s own medical scans.
While more research is needed before clinical use, the idea is promising. A soft, jelly-like material could help heal rock-hard bones. This innovation from ETH Zurich may redefine the future of bone implants and regenerative medicine.