Chemists at the National University of Singapore (NUS) have developed a novel method to convert carbon dioxide—a major greenhouse gas—into valuable liquid hydrocarbons, the primary ingredients in fuels like gasoline and jet fuel.
For years, researchers have been searching for efficient ways to recycle carbon dioxide into energy-rich molecules to reduce emissions and create sustainable fuels. Most of this work has centered on copper catalysts, which can convert carbon dioxide into simple products like ethylene or ethanol. However, copper has consistently struggled to produce longer, branched hydrocarbon chains—essential components of high-performance fuels.
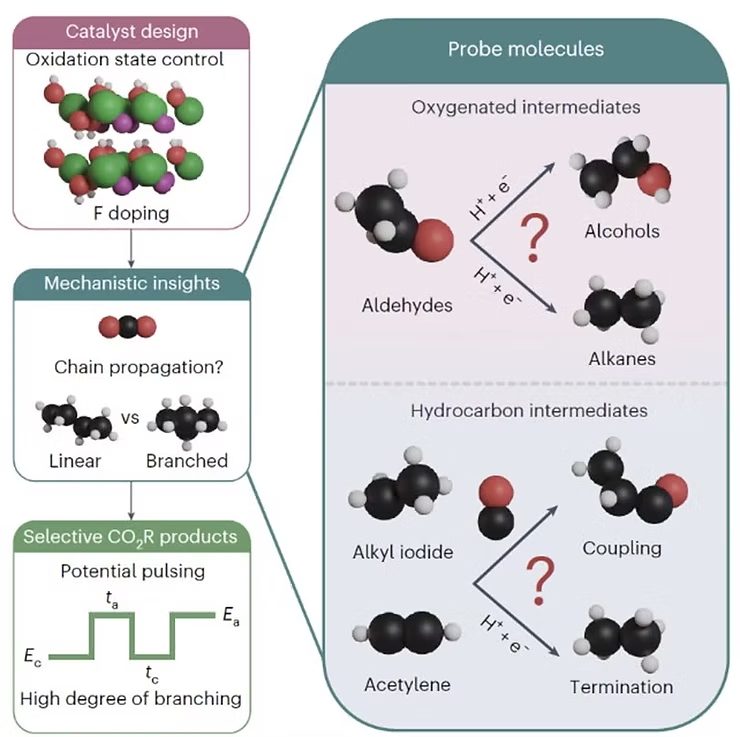
In a new study, a team led by Associate Professor Boon Siang Yeo from NUS’s Department of Chemistry took a different approach. They used a nickel-based catalyst to drive the electrochemical reduction of carbon dioxide. By introducing a small amount of fluoride ions into the nickel structure and using pulsed potential electrolysis, the team was able to fine-tune the reaction.
These innovations gave them remarkable control over the structure of the hydrocarbons produced—specifically, whether the molecules were straight-chained or branched. Branched hydrocarbons are especially desirable for fuel applications because they burn more cleanly and deliver better performance in engines, including those used in cars and airplanes.
The research was conducted in collaboration with Professor Núria López, a specialist in computational simulations at the Institute of Chemical Research of Catalonia in Spain, and Professor Javier Pérez-Ramírez of ETH Zürich in Switzerland, an expert in electro- and thermocatalytic fuel synthesis.
The study introduces innovative strategies to selectively enhance the production of branched hydrocarbons. By using a method known as pulsed potential electrolysis—which involves periodically varying the electrical voltage—the researchers significantly increased the ratio of branched to linear hydrocarbons containing five or more carbon atoms. In fact, they achieved over a 400% improvement compared to conventional techniques. Additionally, doping the nickel catalyst with fluoride ions helped stabilize its oxidation state under reducing conditions, which played a crucial role in supporting the growth of longer hydrocarbon chains.
READ ALSO: Chinese firm develops world’s largest LED mesh Flying Display
While copper-based catalysts have been heavily researched and modified over the past decade, they still struggle to convert carbon dioxide into meaningful amounts of long-chain hydrocarbons. One of the key findings of this study was identifying how nickel catalysts behave differently at the molecular level compared to copper. The team discovered that nickel more effectively removes oxygen from reaction intermediates and encourages asymmetric coupling between absorbed carbon monoxide and unsaturated hydrocarbon species. In contrast, copper tends to convert these intermediates into alcohols, which prevents further chain growth.
This fundamental difference allows nickel catalysts to facilitate the formation and connection of molecular building blocks required for longer, more complex hydrocarbons—closely mimicking the products formed through high-temperature industrial methods like Fischer–Tropsch synthesis.
“This research brings together expertise in catalyst design, reaction mechanism studies, and computational modeling,” said Prof. Yeo. “By combining experimental and theoretical insights, we’ve been able to uncover new pathways and design principles for converting carbon dioxide into long-chain hydrocarbons. Such progress wouldn’t have been possible without close collaboration between experimentalists and theoreticians.”
READ ALSO: The Hutchinson site in Château-Gontier has earned the AeroExcellence Bronze certification.
READ ALSO: Metasurfaces Could Drive the Future of Quantum Information Processing
Dr. Ou Yingqing, a Research Fellow on the team, explained, “One of the key breakthroughs in our study is uncovering why copper-based catalysts—despite years of intense research—struggle to generate significant amounts of long-chain hydrocarbons, unlike their nickel-based counterparts.”
The implications of this research extend well beyond advancing the basic science of carbon dioxide electroreduction. By demonstrating how to precisely control the structure of hydrocarbons produced from CO₂ using electricity, the study lays the groundwork for developing on-demand, sustainable aviation fuels and chemical feedstocks. These advancements play a vital role in accelerating the transition to cleaner, more sustainable energy technologies worldwide.